HR Pharmaceuticals Receives Exclusive Commercialization Rights To Poiesis Medical’s Dual Balloon Catheter Technology In No America
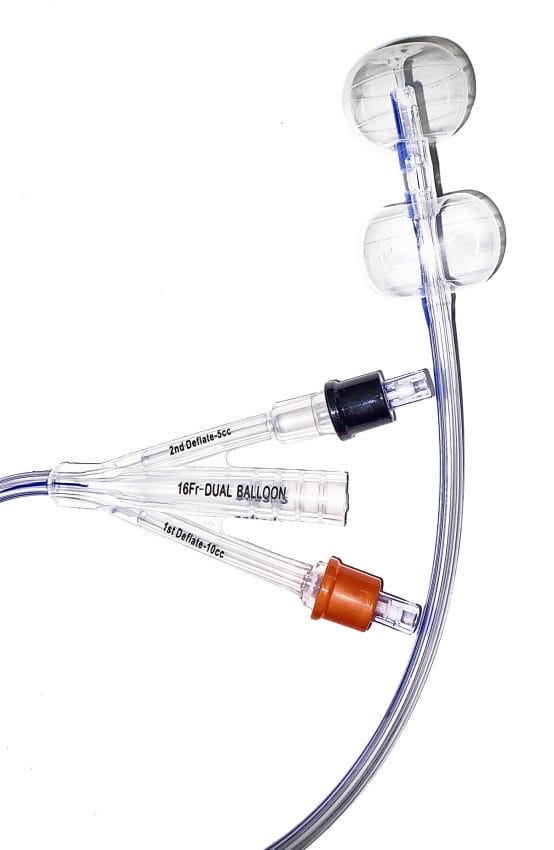
HR Pharmaceuticals, Inc. (HRP) has obtained unique commercialization rights for Poiesis Medical LLC’s Twin Balloon Catheter (Duette™) in North America, additional enhancing HRP’s urological portfolio and addressing adversarial occasions associated to Foley catheterization for particular affected person populations.
The Twin Balloon Catheter has demonstrated important advantages in comparison with conventional Foley catheters, main to raised affected person outcomes. Particularly, we see a terrific alternative for sufferers requiring indwelling catheterization better than 5 days. This product will turn into an essential a part of our broader urology technique over the following 12 months,” states Colby Wiesman, President of HR Prescription drugs, Inc.
“The Twin Balloon Catheter expertise, a brainchild of my father, Dr. Bruce Wiita, a training urologist and adorned Vietnam veteran, was born out of a real want to enhance the standard of life for sufferers enduring extended catheterization and to alleviate the strains it locations on the healthcare system,” says Gregory Wiita, CEO and Founder at Poiesis Medical LLC. “We’re enthusiastic about this new chapter with HR Prescription drugs and the potential of the expertise to make a significant impression in affected person care.”
HRP will start processing North American orders for the Duette Twin Balloon Catheters beginning January 2, 2024. Duette is accessible at most nationwide distributors, together with Cardinal Well being, McKesson, Medline, and Owens & Minor.
Trending Merchandise










